Nematodes show how lack of food shapes the next generation
A study led by the University of Bern provides new insights into how environmental conditions can have a lasting effect over generations. Using nematodes, the researchers were able to show that the nutritional conditions of mother animals significantly influence the early growth of their offspring: If mother animals receive less food, they endow their offspring with fewer ribosomes, the "protein factories" of the cell, and the offspring grow more slowly at first as a result. The study thus opens up new perspectives on non-genetic inheritance.
Whether organisms grow quickly or slowly depends heavily on nutrient availability. If there is little food available, organisms switch to economy mode: they curb their metabolism and invest less energy in growth and in the production of proteins required for growth. Protein production takes place in the "protein factories" of the cell, so-called ribosomes. If fewer nutrients are available, organisms reduce their number of ribosomes. If there are fewer ribosomes, fewer proteins are produced and as a result, the cells grow more slowly and divide less frequently. But how do such adaptations in organisms affect their offspring?
This is the focus of a new study led by Prof. Dr. Benjamin Towbin from the Institute of Cell Biology at the University of Bern, in collaboration with the Centre for Genomic Regulation in Barcelona. Using the tiny nematode Caenorhabditis elegans, the researchers show that the nutritional conditions of mother animals determine how well their offspring grow later on. The study has just been published in PLOS Biology.
Nematodes as a model: new insights into the consequences of food shortages
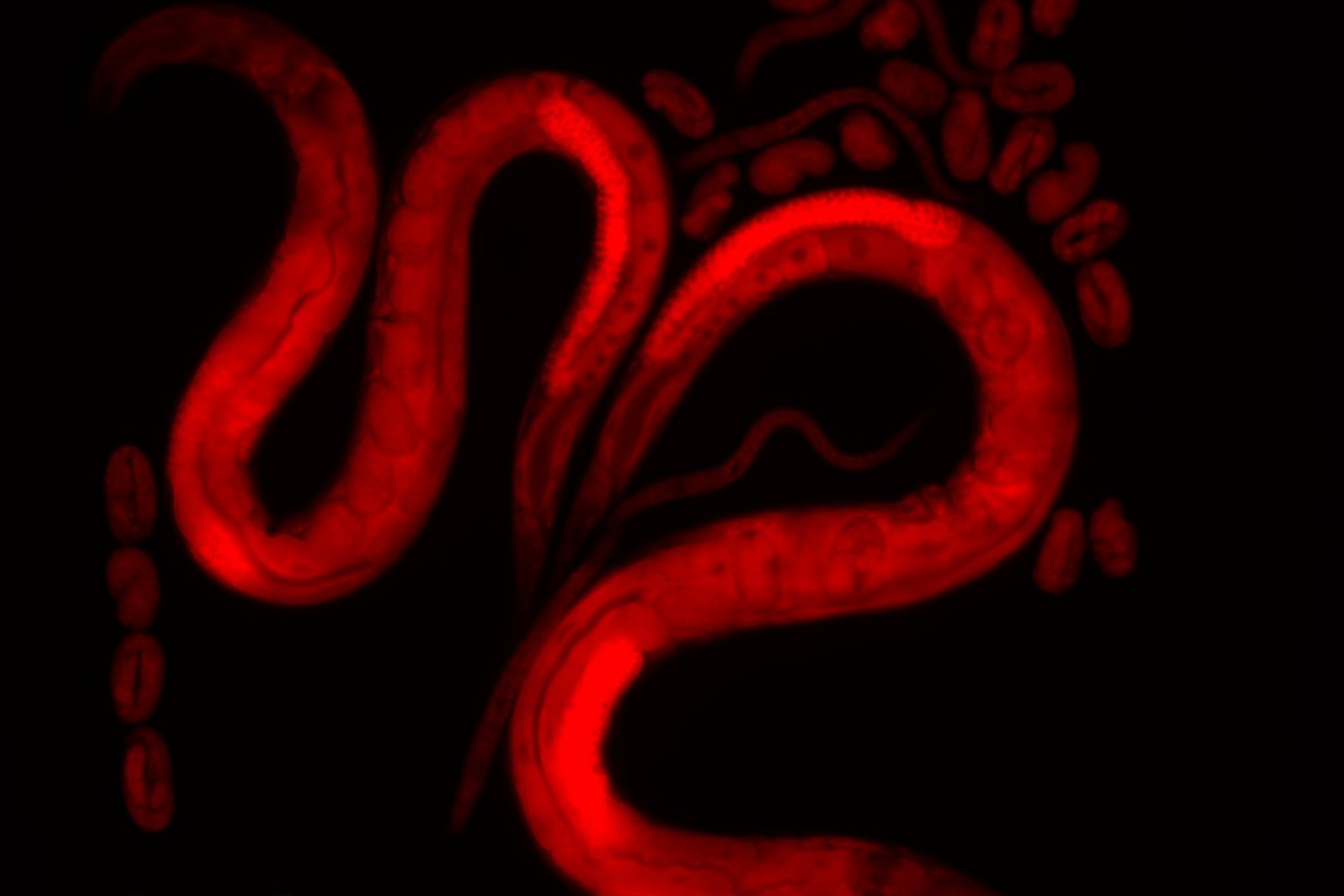
For their study, the research team used the nematode Caenorhabditis elegans, a frequently used model organism in research. "A model organism is a species of animal, plant or microbe that is frequently used in research because it is a good model for studying fundamental biological processes," explains research group leader Benjamin Towbin. Using CRISPR/Cas9 genome editing, the researchers modified the nematodes so that their ribosomes glowed fluorescently.
The researchers then either gave the nematodes less food than normal or fed them without restricting their diet. Using high-resolution fluorescence video microscopy, the team examined the number of ribosomes per cell in both the mother animals and their offspring. This technique makes it possible to record fluorescent structures in living cells or organisms over a period of days and thus observe the development of these structures. In this way, the researchers were able to simultaneously determine how quickly the nematodes grow and how many ribosomes there are in their cells.
Inherited number of ribosomes determines growth
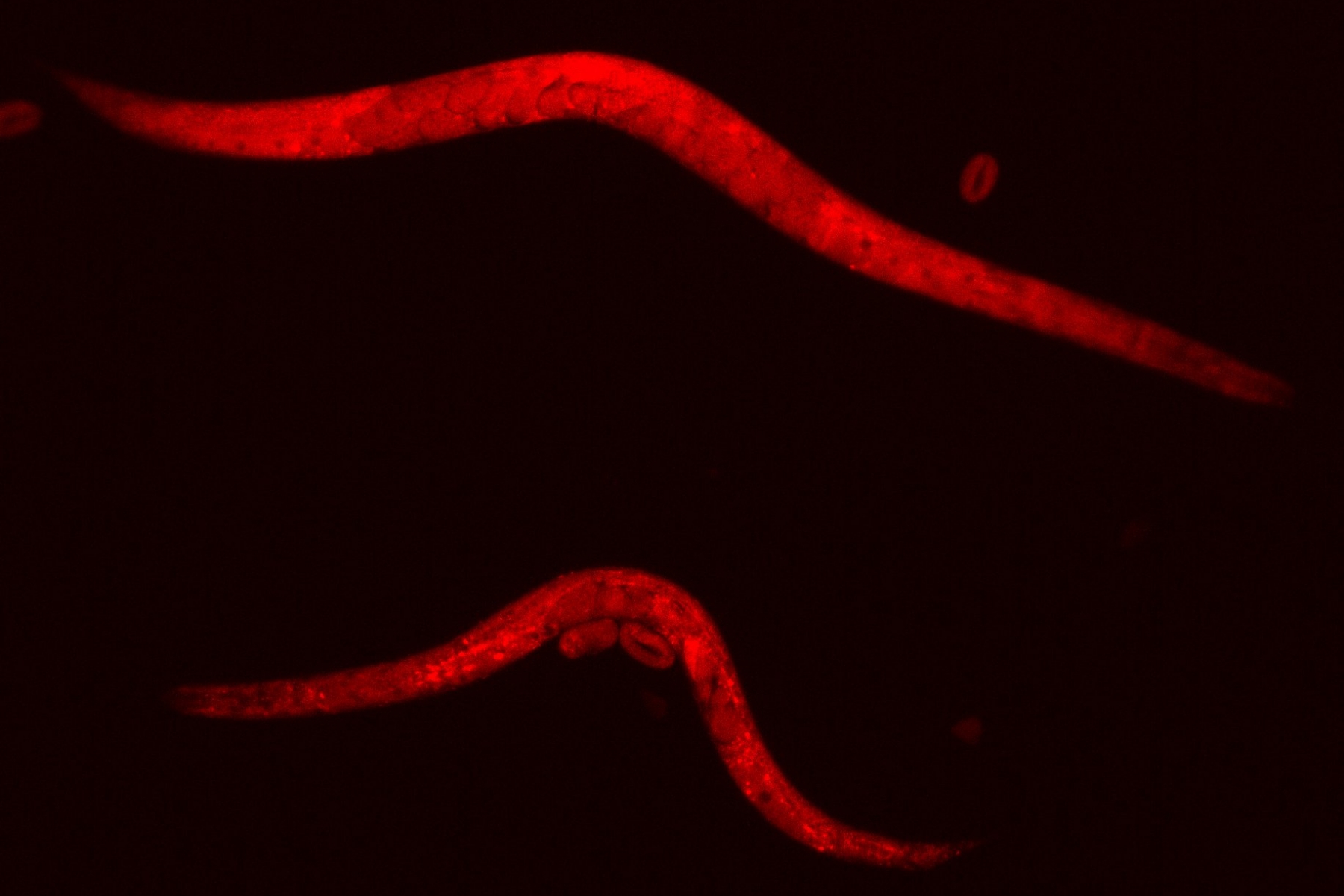
"The nematodes that we provided with a reduced amount of food had a lower number of ribosomes. This was to be expected," explains Towbin. However, the study also shows that the number of ribosomes is significantly reduced in a newly hatched larva if its mother's food supply was previously restricted. "We were therefore able to demonstrate that a nematode passes on its own ribosome content to its offspring," says Sigma Pradhan, first author of the study and former doctoral student at the Institute of Cell Biology at the University of Bern. The researchers managed to show that ribosome composition is passed on by mother animals changing the composition of the developing egg cells depending on their own nutritional conditions. This determines how many ribosomes are produced in the egg and passed on to the offspring.
As a result, the offspring start life with fewer "protein factories", produce fewer proteins required for growth and therefore initially grow more slowly. Later stages of development are therefore achieved with a delay. The number of ribosomes that mothers pass on to their offspring can therefore significantly influence their development and survival success. "Until now, it was assumed that external stimuli, such as hunger or stress, lead to a reaction within the cell via a long chain of interlinked molecular steps," explains Towbin. "Surprisingly, our study now shows that not just such complex pathways are required for offspring to respond to their environmental conditions. A comparatively simple and basic mechanism, namely the number of available ribosomes deposited by the mother in the egg, is sufficient to explain how nutritional conditions affect the offspring," says Pradhan. "If the offspring, unlike their mother, have sufficient food after birth, their number of ribosomes normalizes at some point. As a result, the animals will eventually grow and develop normally again. ".
A growth regulator that occurs in many species
In addition to this discovery, the researchers were able to show through genetic experiments that an important "switch" for growth, the so-called mTORC1 signaling pathway, determines the transfer of the number of ribosomes from the mother animals to their offspring. The researchers were able to flip this switch in mother animals, specifically stopping the mTORC1 signaling pathway. As a result, the offspring had reduced ribosomes, even when sufficient nutrients were available.
Many organisms, from yeasts to humans, possess this switch. In particular, they use it to control how much their cells grow and how they utilize food: To this end, the mTORC1 pathway integrates various signals from the cell and its environment – such as whether sufficient nutrients and energy are available – and decides based on them whether growth is promoted or slowed down. "mTORC1 is therefore a kind of control center that decides whether cells grow," explains Towbin.
"The fact that this signaling pathway is involved in passing on the number of ribosomes from mothers to their offspring and that it is present in many organisms suggests that similar mechanisms could also play a role in other organisms – even though our study was only carried out in nematodes," says Pradhan.
New perspective on non-genetic inheritance
"As this is basic research, it is currently difficult to say to what extent these results can be transferred to other organisms, or even to humans," says Towbin. However, the study opens up a new perspective for understanding non-genetic inheritance and developmental biology in general. "We now know that not only are genes in the genome, i.e. DNA, passed on to the offspring, but also certain characteristics which were shaped by the living conditions of the mother animals – in our case a lack of food and the resulting change in the number of ribosomes," says Pradhan. Towbin continues: "Our results show that such environmental conditions can influence the starting position of the next generation without their genes themselves changing". In a next step, the researchers will investigate whether the production and degradation of ribosomes also explain other important processes, such as surviving periods of famine or ageing.
Publication details:Pradhan, S. et al. (2026). Dietary restriction shapes intergenerational ribosome abundance and early growth of C. elegans offspring, PLOS Biology. |
Institute of Cell BiologyThe Institute of Cell Biology at the University of Bern conducts cutting-edge research in molecular and cell biology to investigate a variety of topics that include host-pathogen interactions, systems biology, epigenetics, organelle biogenesis and developmental processes. |
2026/04/10